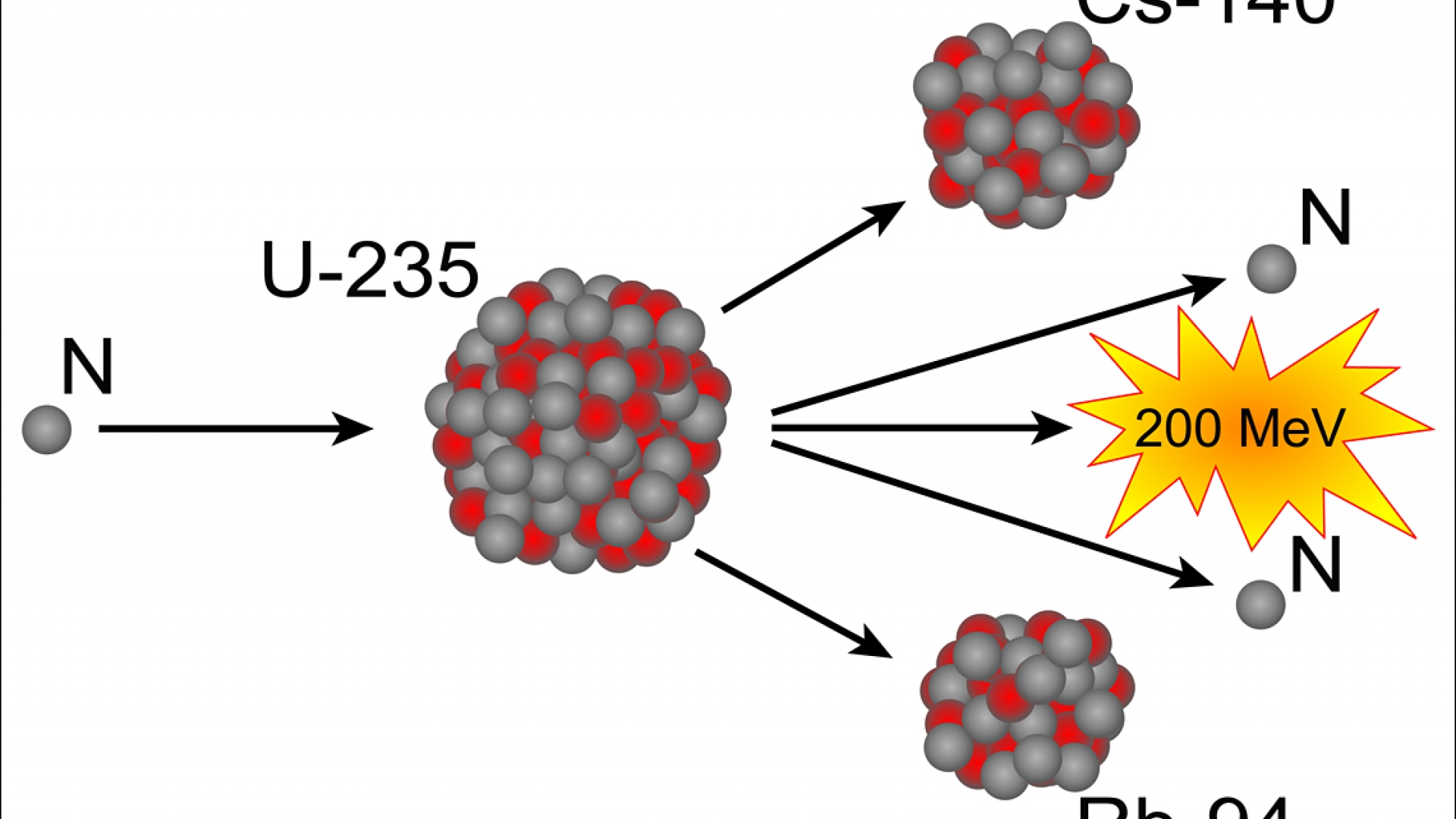
Natural uranium is only about 0.7 U-235, the fissile isotope. Some of them produce neutrons, called delayed neutrons, which contribute to the fission chain reaction. Uranium-235 is commonly used as a fuel in nuclear reactors It has a very long half-life of 700 million years This means that it would have a low activity and. To make uranium fission more efficient, nuclear engineers enrich it. A critical chain reaction can be achieved at low concentrations of U-235 if the neutrons from fission are moderated to lower their speed, since the probability for fission with slow neutrons is greater.Ī fission chain reaction produces intermediate mass fragments which are highly radioactive and produce further energy by their radioactive decay. If the reaction will sustain itself, it is said to be "critical", and the mass of U-235 required to produced the critical condition is said to be a " critical mass". 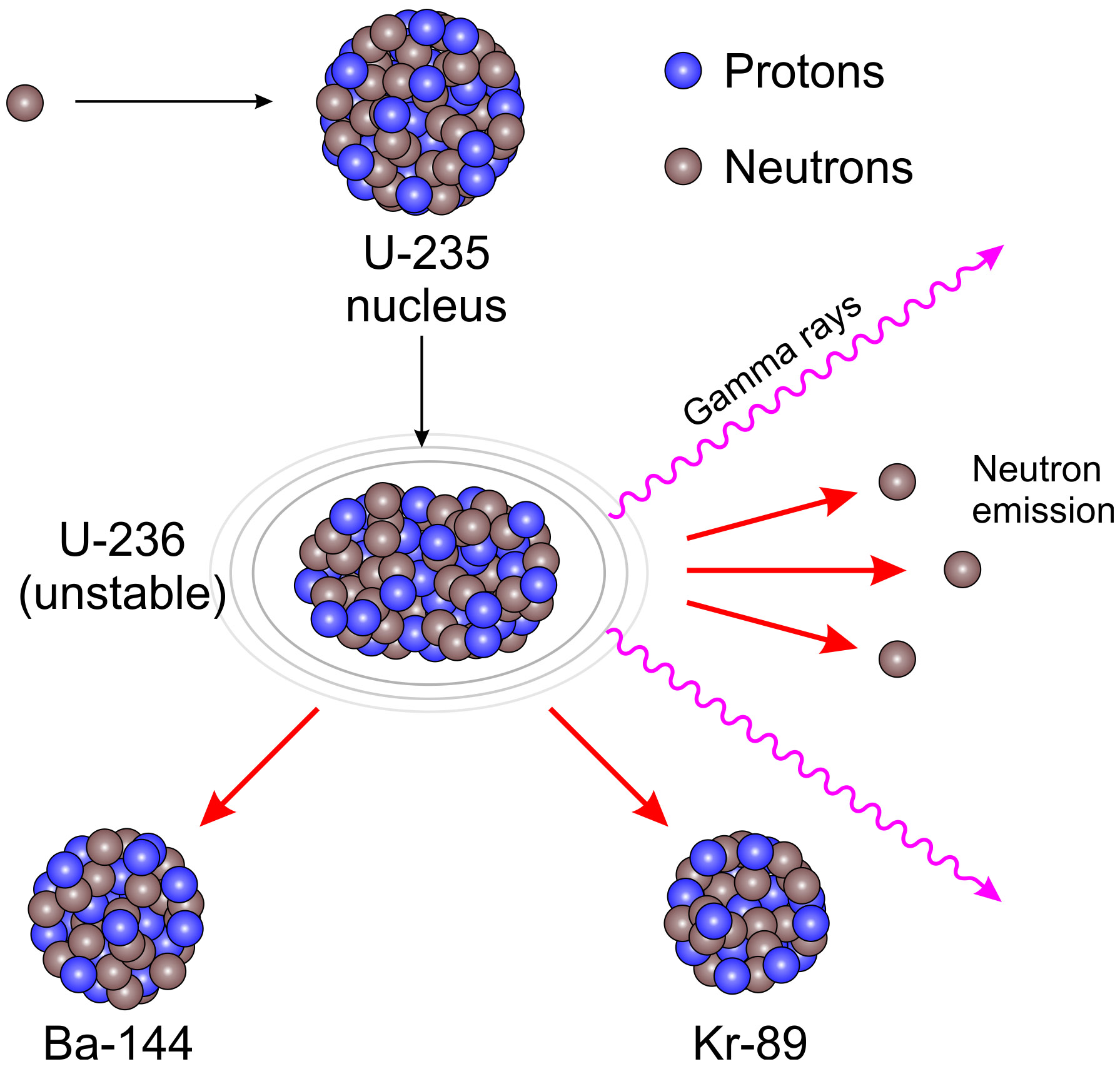
If an least one neutron from U-235 fission strikes another nucleus and causes it to fission, then the chain reaction will continue. If at least one neutron from each fission strikes another U-235 nucleus and initiates fission, then the chain reaction is sustained. It was December 1938 when the radiochemists Otto Hahn (above, with Lise Meitner) and Fritz Strassmann, while bombarding elements with neutrons in their Berlin. These fragments, or fission products, are about equal to half the. Uranium-235 Fission Example Initiation of this processĮnergy From Uranium Fission Form of Energy ReleasedĮnergy of decay products of fission fragments This type of the fission caused by fast neutron bombardment is mainly composed of the 5050 splitting of a uranium atom, and is called symmetric fission. When a nucleus fissions, it splits into several smaller fragments. Uranium-235 Chain Reaction Uranium-235 Fission Detailed example
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
